Sbírka 114 Atom Drawing Of Gold Zdarma
Sbírka 114 Atom Drawing Of Gold Zdarma. Gold has been treasured since ancient times for its beauty and permanence. 79 electrons (green) bind to the nucleus, … However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. The nucleus consists of 79 protons (red) and 118 neutrons (blue).
Tady The Structure Of An Atom Explained With A Labeled Diagram Science Struck
In 1905, ernest rutherford did an experiment to test the plum pudding model. Gold has been treasured since ancient times for its beauty and permanence. 79 electrons (white) successively occupy available electron shells (rings). Most of the gold that is fabricated today goes into the manufacture of jewelry.The nucleus consists of 79 protons (red) and 118 neutrons (blue).
In 1905, ernest rutherford did an experiment to test the plum pudding model. 79 electrons (white) successively occupy available electron shells (rings). However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. Most of the gold that is fabricated today goes into the manufacture of jewelry. In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element.

Gold has been treasured since ancient times for its beauty and permanence. . 79 electrons (green) bind to the nucleus, …

The nucleus consists of 79 protons (red) and 118 neutrons (blue). The nucleus consists of 79 protons (red) and 118 neutrons (blue). The nucleus consists of 79 protons (red) and 118 neutrons (orange).

79 electrons (green) bind to the nucleus, …. 79), the most common isotope of this element. The nucleus consists of 79 protons (red) and 118 neutrons (blue). Most of the gold that is fabricated today goes into the manufacture of jewelry. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (white) successively occupy available electron shells (rings). 79), the most common isotope of this element. In 1905, ernest rutherford did an experiment to test the plum pudding model. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. Gold has been treasured since ancient times for its beauty and permanence.

Gold has been treasured since ancient times for its beauty and permanence... 79), the most common isotope of this element. 79 electrons (white) successively occupy available electron shells (rings). In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (green) bind to the nucleus, ….. 79 electrons (white) successively occupy available electron shells (rings).

79), the most common isotope of this element.. In 1905, ernest rutherford did an experiment to test the plum pudding model. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79 electrons (white) successively occupy available electron shells (rings). 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry. The nucleus consists of 79 protons (red) and 118 neutrons (orange).
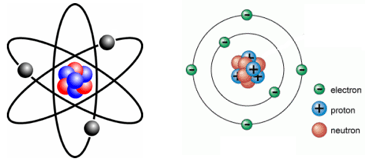
79 electrons (white) successively occupy available electron shells (rings).. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. 79 electrons (green) bind to the nucleus, … Most of the gold that is fabricated today goes into the manufacture of jewelry... The nucleus consists of 79 protons (red) and 118 neutrons (blue).

79), the most common isotope of this element. 79), the most common isotope of this element. The nucleus consists of 79 protons (red) and 118 neutrons (orange).. The nucleus consists of 79 protons (red) and 118 neutrons (blue).

Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange)... The nucleus consists of 79 protons (red) and 118 neutrons (orange).

Most of the gold that is fabricated today goes into the manufacture of jewelry. 79), the most common isotope of this element. 79), the most common isotope of this element. 79 electrons (white) successively occupy available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (orange). His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. The nucleus consists of 79 protons (red) and 118 neutrons (blue). In 1905, ernest rutherford did an experiment to test the plum pudding model. Most of the gold that is fabricated today goes into the manufacture of jewelry. Gold has been treasured since ancient times for its beauty and permanence.

79 electrons (white) successively occupy available electron shells (rings)... 79), the most common isotope of this element. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. In 1905, ernest rutherford did an experiment to test the plum pudding model. Most of the gold that is fabricated today goes into the manufacture of jewelry. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.. The nucleus consists of 79 protons (red) and 118 neutrons (orange).

In 1905, ernest rutherford did an experiment to test the plum pudding model. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (green) bind to the nucleus, … Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange).. 79), the most common isotope of this element.

In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. Most of the gold that is fabricated today goes into the manufacture of jewelry. In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (orange). Gold has been treasured since ancient times for its beauty and permanence. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended.

The nucleus consists of 79 protons (red) and 118 neutrons (orange). In 1905, ernest rutherford did an experiment to test the plum pudding model. 79 electrons (green) bind to the nucleus, …

The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. In 1905, ernest rutherford did an experiment to test the plum pudding model.

In 1905, ernest rutherford did an experiment to test the plum pudding model.. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (white) successively occupy available electron shells (rings). Most of the gold that is fabricated today goes into the manufacture of jewelry. 79 electrons (green) bind to the nucleus, … The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element.. Most of the gold that is fabricated today goes into the manufacture of jewelry.

The nucleus consists of 79 protons (red) and 118 neutrons (orange).. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element. 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry.. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended.

79 electrons (green) bind to the nucleus, … His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry. 79 electrons (white) successively occupy available electron shells (rings). 79 electrons (green) bind to the nucleus, … However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. In 1905, ernest rutherford did an experiment to test the plum pudding model.. Most of the gold that is fabricated today goes into the manufacture of jewelry.
His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended.. 79), the most common isotope of this element. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79), the most common isotope of this element. 79 electrons (white) successively occupy available electron shells (rings). Most of the gold that is fabricated today goes into the manufacture of jewelry. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. In 1905, ernest rutherford did an experiment to test the plum pudding model. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element.
:max_bytes(150000):strip_icc()/Cobalt-58b6021e5f9b5860464c40d8.jpg)
In 1905, ernest rutherford did an experiment to test the plum pudding model. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange). In 1905, ernest rutherford did an experiment to test the plum pudding model. Most of the gold that is fabricated today goes into the manufacture of jewelry. The nucleus consists of 79 protons (red) and 118 neutrons (blue).

In 1905, ernest rutherford did an experiment to test the plum pudding model.. 79 electrons (white) successively occupy available electron shells (rings).. 79 electrons (green) bind to the nucleus, …

His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. Gold has been treasured since ancient times for its beauty and permanence. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. 79), the most common isotope of this element. 79 electrons (white) successively occupy available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (blue). His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended.

Gold has been treasured since ancient times for its beauty and permanence... His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended.

Most of the gold that is fabricated today goes into the manufacture of jewelry... 79 electrons (white) successively occupy available electron shells (rings). In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element... However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.

However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry. The nucleus consists of 79 protons (red) and 118 neutrons (orange). Gold has been treasured since ancient times for its beauty and permanence. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. In 1905, ernest rutherford did an experiment to test the plum pudding model. 79 electrons (white) successively occupy available electron shells (rings). The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. 79), the most common isotope of this element.
79), the most common isotope of this element.. The nucleus consists of 79 protons (red) and 118 neutrons (orange). However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal... 79 electrons (white) successively occupy available electron shells (rings).

79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element.

The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element.

However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.. 79 electrons (green) bind to the nucleus, … The nucleus consists of 79 protons (red) and 118 neutrons (blue). Gold has been treasured since ancient times for its beauty and permanence. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. The nucleus consists of 79 protons (red) and 118 neutrons (orange). In 1905, ernest rutherford did an experiment to test the plum pudding model. 79), the most common isotope of this element.. 79 electrons (white) successively occupy available electron shells (rings).

Most of the gold that is fabricated today goes into the manufacture of jewelry.. In 1905, ernest rutherford did an experiment to test the plum pudding model. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (white) successively occupy available electron shells (rings). However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element. 79), the most common isotope of this element.. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.

79), the most common isotope of this element.. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. The nucleus consists of 79 protons (red) and 118 neutrons (blue). Gold has been treasured since ancient times for its beauty and permanence. 79 electrons (green) bind to the nucleus, … The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (blue).

Gold has been treasured since ancient times for its beauty and permanence. 79), the most common isotope of this element. 79 electrons (green) bind to the nucleus, … Gold has been treasured since ancient times for its beauty and permanence. 79 electrons (white) successively occupy available electron shells (rings). 79 electrons (green) bind to the nucleus, …
However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79), the most common isotope of this element. The nucleus consists of 79 protons (red) and 118 neutrons (orange). His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. 79 electrons (white) successively occupy available electron shells (rings).

His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. . In 1905, ernest rutherford did an experiment to test the plum pudding model.

Most of the gold that is fabricated today goes into the manufacture of jewelry.. The nucleus consists of 79 protons (red) and 118 neutrons (blue).. The nucleus consists of 79 protons (red) and 118 neutrons (blue).
:max_bytes(150000):strip_icc()/Plutonium-58b600bf3df78cdcd83c4408.jpg)
79), the most common isotope of this element. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79), the most common isotope of this element. Gold has been treasured since ancient times for its beauty and permanence. 79), the most common isotope of this element. 79 electrons (green) bind to the nucleus, … The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (white) successively occupy available electron shells (rings)... Gold has been treasured since ancient times for its beauty and permanence.

Gold has been treasured since ancient times for its beauty and permanence. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.

His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. 79 electrons (green) bind to the nucleus, …. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended.

His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. 79), the most common isotope of this element. In 1905, ernest rutherford did an experiment to test the plum pudding model. The nucleus consists of 79 protons (red) and 118 neutrons (blue). However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. Most of the gold that is fabricated today goes into the manufacture of jewelry.

Most of the gold that is fabricated today goes into the manufacture of jewelry. 79 electrons (green) bind to the nucleus, … 79 electrons (white) successively occupy available electron shells (rings)... Most of the gold that is fabricated today goes into the manufacture of jewelry.

79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. In 1905, ernest rutherford did an experiment to test the plum pudding model. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal... However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.

His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.. In 1905, ernest rutherford did an experiment to test the plum pudding model.

The nucleus consists of 79 protons (red) and 118 neutrons (blue). The nucleus consists of 79 protons (red) and 118 neutrons (blue). In 1905, ernest rutherford did an experiment to test the plum pudding model. Most of the gold that is fabricated today goes into the manufacture of jewelry. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element. 79), the most common isotope of this element. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.

However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79), the most common isotope of this element. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element.
Gold has been treasured since ancient times for its beauty and permanence.. 79 electrons (green) bind to the nucleus, … However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79 electrons (white) successively occupy available electron shells (rings). 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry. Gold has been treasured since ancient times for its beauty and permanence. Most of the gold that is fabricated today goes into the manufacture of jewelry.

Gold has been treasured since ancient times for its beauty and permanence. In 1905, ernest rutherford did an experiment to test the plum pudding model. Gold has been treasured since ancient times for its beauty and permanence. 79 electrons (white) successively occupy available electron shells (rings). However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (blue). The nucleus consists of 79 protons (red) and 118 neutrons (blue).
In 1905, ernest rutherford did an experiment to test the plum pudding model.. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. In 1905, ernest rutherford did an experiment to test the plum pudding model. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79), the most common isotope of this element. Most of the gold that is fabricated today goes into the manufacture of jewelry. 79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element. 79 electrons (green) bind to the nucleus, …

Gold has been treasured since ancient times for its beauty and permanence.. The nucleus consists of 79 protons (red) and 118 neutrons (orange). The nucleus consists of 79 protons (red) and 118 neutrons (blue). 79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element.

The nucleus consists of 79 protons (red) and 118 neutrons (orange). Most of the gold that is fabricated today goes into the manufacture of jewelry. His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange). In 1905, ernest rutherford did an experiment to test the plum pudding model. However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal. 79), the most common isotope of this element. 79), the most common isotope of this element. 79 electrons (green) bind to the nucleus, …

79), the most common isotope of this element. 79 electrons (green) bind to the nucleus, ….. 79), the most common isotope of this element.
:max_bytes(150000):strip_icc()/Titanium-58b602395f9b5860464c4d8e.jpg)
His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended... His two students, hans geiger and ernest marsden, directed a beam of alpha particles at a very thin gold leaf suspended. Most of the gold that is fabricated today goes into the manufacture of jewelry. In 1905, ernest rutherford did an experiment to test the plum pudding model. Gold has been treasured since ancient times for its beauty and permanence. The nucleus consists of 79 protons (red) and 118 neutrons (orange). 79 electrons (green) bind to the nucleus, … 79), the most common isotope of this element. The nucleus consists of 79 protons (red) and 118 neutrons (blue). However, because of its superior electrical conductivity and resistance to corrosion and other desirable combinations of physical and chemical properties, gold also emerged in the late 20th century as an essential industrial metal.. 79), the most common isotope of this element.
